Materials and Methodsįor this quality assurance project, we retrospectively identified 245 patients with primary invasive breast cancer who received NAC at the Breast Center, Department of OB&GYN, LMU University Hospital, Munich, Germany. Therefore, this study aimed to assess the incidence of a switch in HR and HER2 status after state-of-the-art NAC in primary invasive breast cancer. We hypothesized that a relevant number of cases with primary invasive breast cancer present with a switch of hormone receptor (HR) and/or HER2 status in the non-pCR breast cancer specimen after neoadjuvant chemotherapy (NAC). Correspondingly, the German AGO breast commission sees reevaluation of ER, PgR, and HER2 in case of non-pCR as an optional procedure. Gallen consensus did not explicitly vote on biomarker re-testing. Gallen consensus ) do not recommend general biomarker re-evaluation. Current guidelines (ESMO, ASCO, NCCN, St. However, pCR rates vary widely based on various characteristics. In specific subtypes (HER2-positive and triple-negative breast cancers ), neoadjuvant treatment has become the standard of care as pCR is closely correlated with patient outcome and adjuvant therapy choices may differ based on the pCR status. Pathological complete response (pCR) defined as no invasive tumor in breast and lymph nodes (ypT0/is ypN0) has an impact on subsequent patient outcome. These markers are recognized by international guidelines as predictive factors and are thus indispensable for therapy decision-making. Determination of estrogen receptor (ER), progesterone receptor (PgR), and HER2 status is mandatory for all patients with invasive breast cancer. Neoadjuvant application is preferred for response-guided systemic and local therapy, and if tumor size reduction is warranted for optimal surgical results or to assess the tumor’s response in vivo. Chemotherapy can be given before (neoadjuvant) or after (adjuvant) surgery with equivalent effects on outcomes, as first demonstrated by the NSABP-B18 trial.  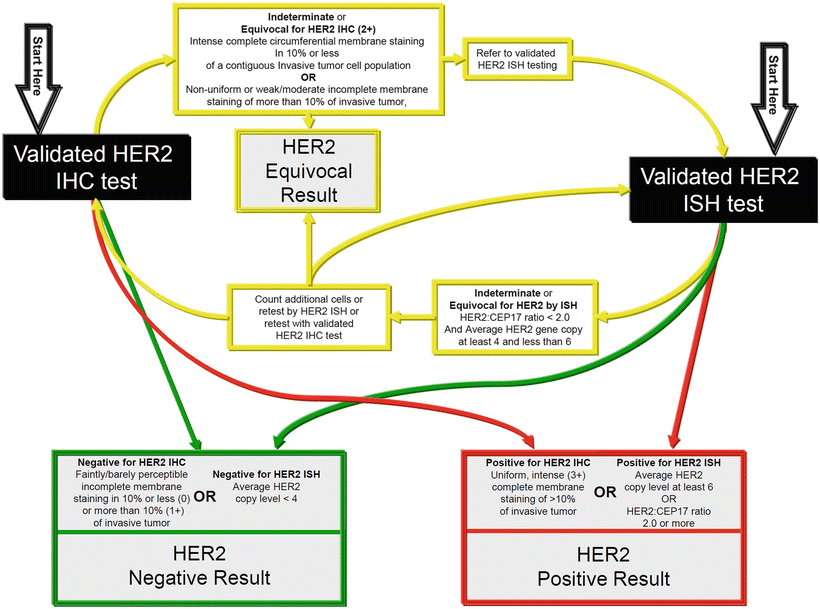
Systemic therapies for early breast cancer are highly effective the individual indication depends on patient characteristics, molecular subtype, tumor burden, and absolute risk of recurrence. Particularly, in view of the recent therapy advances, it will be critical to evaluate whether individualization of treatment concepts based on the biology of the non-pCR specimens is preferable to the initial therapy concept based on the pathology at primary diagnosis. Future research needs to address the impact of HR and HER2 status switch on therapy adaptation and on subsequent patient outcome. Yet, routine reevaluation of HR and HER2 status is not recommended in guidelines so far. These results are consistent with prior evidence. Conclusion: In a substantial number (27%) of non-pCR cases, a switch in biomarker status after completed neoadjuvant treatment was detected. Twenty cases had a switch in HR status, while 15 cases had a switch in HER2 status. In 35 cases (27%), a switch of either HR and/or HER2 status between the initial biopsy and the surgical specimen was detected. In 128 patients (52%), surgery revealed non-pCR after completed NAC. Results: 245 consecutive patients with primary invasive breast cancer, treated with NAC with/without targeted therapy between Januand December 31, 2019, at the LMU Breast Center, Munich, Germany, were identified. HR and HER2 status determined before NAC was compared with the corresponding receptor status determined in the surgical specimen after NAC. Pathological complete response (pCR) was defined as no invasive tumor in breast and lymph nodes (ypT0/is ypN0). Methods: Tumor samples from patients without pathological complete response (non-pCR) were evaluated. Introduction: This project aimed to identify the frequency of a switch of hormone receptor (HR) and/or HER2 status after neoadjuvant chemotherapy (NAC) for early breast cancer. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
